Unauthorized reproduction or distribution is illegal and subject to criminal penalties.
Pharm Edaily enforces a zero-tolerance policy and will take strict action.
[Seok Ji-hoen, Edaily reporter] South Korean biotech stocks rose on Oct. 10 as companies with strong R&D momentum and global catalysts attracted investor attention. AprilBio climbed more than 12% on news that its U.S. partner filed for an initial public offering (IPO). Onconic Therapeutics hit the daily upper limit following news tied to its new drug candidate and a recent stock split. Toolgen also gained over 13% amid expectations for imminent U.S. patent approval.
Pipeline Value Expected to Rise According to MP Doctor (formerly Market Point) operated by KG Zeroin, shares of AprilBio closed up 12.07% at 34,350 won. The rally followed reports that its global partner Evommune has filed for an IPO on the New York Stock Exchange (NYSE).
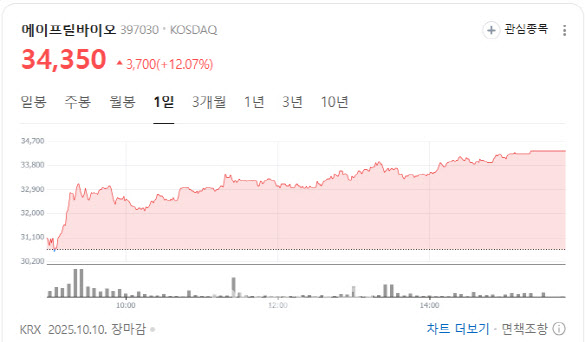 | | (Screenshot: Naver Finance) |
|
Earlier in the day, Edaily reported through its premium content service that Evommune’s IPO plans could boost AprilBio’s pipeline valuation and milestone revenue potential.
AprilBio licensed out its autoimmune disease treatment candidate APB-R3 (EVO301) to Evommune in June 2024 in a deal worth up to $475 million (about 657 billion won). EVO301 is a long-acting fusion protein that selectively inhibits the IL-18 inflammatory pathway and is currently in Phase 2 clinical trials for moderate-to-severe atopic dermatitis. Evommune has also designated ulcerative colitis (UC) as the next indication and is preparing to initiate trials.
Industry analysts say Evommune’s high valuation upon listing could elevate AprilBio’s global recognition and expected milestone payments.
“If Evommune earns a strong market valuation in the U.S., it would further validate our pipeline’s global competitiveness and increase the likelihood of milestone realization,” an AprilBio official said.
“Patent Approval Imminent” Toolgen shares closed up 13.18% at 68,700 won. The stock rose amid expectations that the company’s U.S. patent for Cas9/RNA complex-based gene-editing technology will be registered soon after Toolgen recently paid the Utility Issue Fee to the U.S. Patent and Trademark Office (USPTO).
 | | (Screenshot: Naver Finance) |
|
According to Edaily’s exclusive report, Toolgen submitted the payment on Sept. 30 for its patent titled “Cas9/RNA Complexes for Inducing Modifications of Target Endogenous Nucleic Acid Sequences in Nuclei of Eukaryotic Cells.” The move effectively confirms patent registration, with a patent number expected to be issued shortly.
The patent covers a gene-editing method using a Cas9 protein and guide RNA complex, which enables precise modification of genetic sequences inside cells. Industry observers note that the inclusion of the RNP (ribonucleoprotein) approach?a clinically practical technology?adds significant commercial potential for future gene-editing therapies.
Meanwhile, Toolgen said there were no undisclosed material facts behind its recent share price volatility. In response to a Korea Exchange inquiry on Oct. 2 regarding “unusual trading movements,” the company officially stated on Oct. 10 that “there are no matters requiring disclosure under Article 6 of KOSDAQ disclosure regulations.”
Stock Split and Drug Optimism Lift Onconic Onconic Therapeutics soared 29.75% to 21,150 won, hitting the daily limit-up. Analysts attributed the sharp rise to a mix of optimism surrounding its oncology drug candidate Nesuparib and the technical effect of a recent stock split (bonus issue).
 | | (Screenshot: Naver Finance) |
|
According to the company, results from preclinical studies of Nesuparib in pancreatic cancer were recently published in the International Journal of Biological Sciences (IJBS), a top-tier life science journal.
The study, led by Seoul National University’s Prof. Oh Do-yeon, showed that Nesuparib’s dual inhibition of Tankyrase and PARP enzymes blocked cancer cell growth and DNA repair, potentially overcoming the limitations of existing PARP inhibitors. The candidate demonstrated roughly 10 times stronger cell-growth inhibition than Olaparib in BRCA2-deficient pancreatic cancer cells and efficacy even in BRCA wild-type cases.
“These results support Nesuparib’s potential as a first-line pancreatic cancer therapy and could positively influence ongoing out-licensing discussions,” an Onconic spokesperson said.
Onconic also carried out a bonus share issue last month, granting three new shares for each existing one. The ex-rights date was Oct. 10, meaning only shareholders of record through the previous trading day were eligible for the new shares.
“Despite the ex-rights adjustment, we believe investors’ confidence in the company’s long-term value contributed to today’s share price movement,” the company added.