Unauthorized reproduction or distribution is illegal and subject to criminal penalties.
Pharm Edaily enforces a zero-tolerance policy and will take strict action.
[Yu Jin-hee, Edaily Reporter] On the 26th, the South Korean biotech sector took the baton from the semiconductor rally, recording explosive growth. As news of innovative clinical data and trillion-won global contracts broke, investor sentiment shifted rapidly from “mid-risk, high-return” to high-conviction growth stocks. Notably, three companies—Intron Bio, Gencurix, and SAMCHUNDANG PHARM—hit the daily upper limit, leading the sector‘s surge.
According to KG Zeroin MP DOCTOR, Intron Bio, Gencurix, and SAMCHUNDANG PHARM were listed among the “Top 10 Gainers” in the domestic market. Their stock prices surged by 30.00% (KRW 4,615), 29.97% (KRW 4,510), and 29.85% (KRW 757,000), respectively, proving that the biotech sector is becoming a powerful theme rivaling semiconductors.
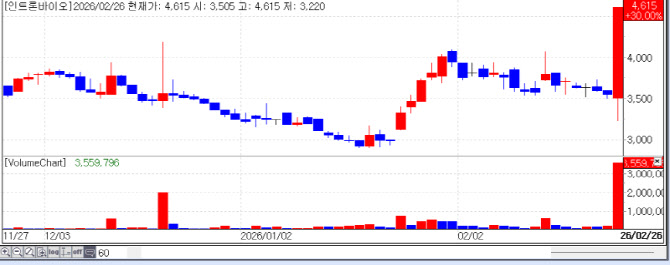 | | Recent stock price trend of Intron Bio. (Source: KG Zeroin MP DOCTOR) |
|
◇ Intron Bio: Poised to Dominate the Superbug Market with ’Complete Sterilization‘ Data Intron Bio received strong market support following news of its upcoming data presentation for ’SAL200‘ at the AMR Conference 2026 in Switzerland next month. The primary driver for the surge is the groundbreaking ’complete sterilization‘ data.
SAL200 is a novel drug derived from bacteriophages, utilizing the Endolysin protein. It features a mechanism of Enzymatic Hydrolysis that immediately dissolves the bacterial cell wall. At the conference, the company will share results showing that SAL200, compared to the standard of care (Daptomycin), induces immediate bacterial lysis, especially in challenging rabbit endocarditis models where it reduced bacterial concentration below the limit of detection—achieving a ’complete sterilization‘ state.
This indicates that SAL200 can overcome the Biofilm environment, a major cause of treatment failure and recurrence. Intron Bio is expected to see a valuation re-rating this year. With SAL200 ready for U.S. FDA Phase 2b and potential licensing-out (L/O) deals with big pharma, the company is also expanding its pipeline through platforms like ’itLysin‘ and ’IMPA‘. Having established a dedicated GMP facility at its headquarters in Seongnam, the company is transitioning from a research-oriented venture to a comprehensive biotech firm capable of manufacturing and validation.
An official from Intron Bio stated: “We will actively introduce our innovative Endolysin engineering platforms and others at this event. Based on this, we aim to achieve tangible results, such as building joint development partnerships.”
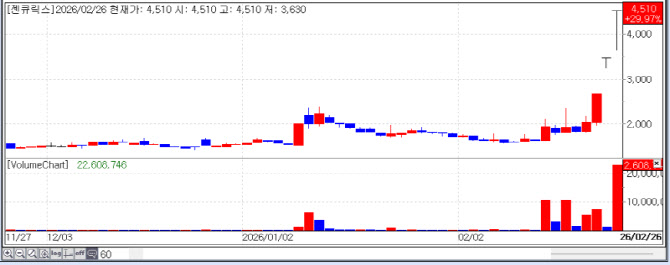 | | Recent stock price trend of Gencurix. (Source: KG Zeroin MP DOCTOR) |
|
◇ Gencurix: The Liquid Biopsy Leader Breaking the ’Pseudo-resistance‘ Barrier Gencurix, a specialist in liquid biopsy-based molecular diagnostics, continued its upward trend by identifying a new cause of drug resistance in cancer and formalizing technical cooperation with the global diagnostic giant Roche.
The first catalyst was the research conducted with Hyundai ADM. The team discovered that pancreatic cancer resistance is often not due to mutations in the cancer cells themselves, but a ’Pseudo-resistance‘ caused by physical barriers in the tumor microenvironment. Gencurix’s high-sensitivity Digital Polymerase Chain Reaction (ddPCR) technology played a crucial role in the precise molecular analysis. The second boost came from being selected for a government R&D project to develop companion diagnostic panels for NSCLC and breast cancer based on Roche‘s ddPCR platform.
Based on this, Gencurix plans to expand its actual revenue. The company holds a portfolio covering the entire cancer cycle, led by ’GensWell BCT‘. Its business model targeting the Japanese lung cancer panel market through PMDA approval and insurance listing ensures substantial growth. With the acquisition of stakes in JinoBio to advance Circulating Tumor Cell (CTC) technology, its platform scalability is expected to grow.
A Gencurix official commented: “The government project selection is business-oriented, targeting cancer types with clear market sizes. We will realize continuous revenue growth in the digital PCR field by enhancing technical reliability through cooperation with a global platform.”
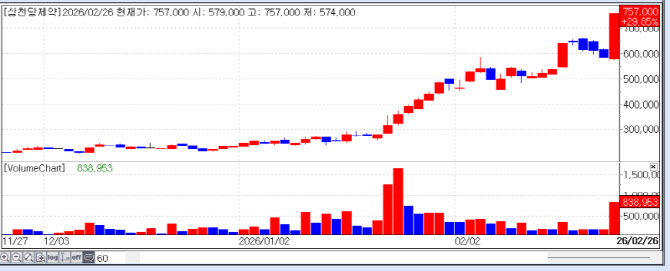 | | Recent stock price trend of SAMCHUNDANG PHARM. (Source: KG Zeroin MP DOCTOR) |
|
◇ SAMCHUNDANG PHARM: A Trillion-Won Jackpot and Game Changer in the Obesity Market SAMCHUNDANG PHARM became a major highlight by signing a KRW 5.3 trillion exclusive licensing and commercialization deal with a global pharmaceutical company in Europe for its oral GLP-1 (Semaglutide) generic.
The contract covers 11 European countries, including the UK and the Netherlands. The core of this deal is the ’SNAC-Free‘ formulation technology developed by SAMCHUNDANG PHARM. This technology perfectly bypasses the original product’s patents while maintaining production costs at approximately 10% of the selling price. Despite being a tender-based market in Europe, the deal features a rare profit-sharing structure where the company receives 60% of net profits. With upfront and milestone payments of approximately KRW 50.8 billion, its financial health has been significantly strengthened.
The European Semaglutide market is predicted to reach KRW 30 trillion by the time of product launch. SAMCHUNDANG PHARM is observed to play a significant role in this market. In fact, following the current 11 countries, contracts with core EU markets such as Germany, France, and Italy are imminent.
A SAMCHUNDANG PHARM official emphasized: “The 60% profit-sharing condition proves our technology‘s ability to bypass patents and its overwhelming cost competitiveness. We are confident in securing a dominant position in major European markets and maximizing revenue.”




![Genome&Company Jumps on Clinical Momentum...ViGenCell Cools [K-Bio Pulse]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/04/PS26043000451b.jpg)



![지놈앤컴퍼니, 임상 본격화 기대감에 급등…숨고르기 들어간 바이젠셀[바이오 맥짚기]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/04/PS26043000447b.jpg)