Unauthorized reproduction or distribution is illegal and subject to criminal penalties.
Pharm Edaily enforces a zero-tolerance policy and will take strict action.
[Yu Jin-hee, Edaily Reporter] Despite uncertainties in the global macroeconomic environment, the domestic stock market has been cruising on a steady upward trajectory. In particular, the biotech sector, centered on the KOSDAQ, is establishing itself as a market leader by delivering tangible clinical data and “earnings surprise” results that go beyond simple speculation.
In contrast to the past, when investor sentiment was dampened by clinical trial suspensions or opaque accounting issues, the recent rally is being driven by “performance-oriented biotech” companies proving their worth through concrete figures. On the 27th, three companies TiumBio, ST Pharm, and ABL Bio saw their stock prices soar by nearly double digits, signaling a full-scale revival of the biotech sector.
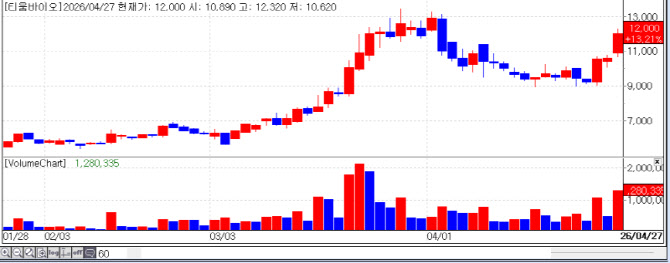 | | Recent stock price trend of TiumBio. (Source: KG Zeroin MP DOCTOR) |
|
TiumBio Shows Unprecedented Pain Relief “Countdown to Global Big Deal” TiumBio skyrocketed 13.21% to close at 12,000 won on the 27th. The market is focusing on the commercialization potential of the company‘s two core pipelines the immuno-oncology candidate ’Tusporsertib‘ (TU2218) and the endometriosis treatment ’Merigolix‘ (TU2670).
First, Tusporsertib proved its technological prowess as the abstract for its Phase 2a interim results (in combination with Keytruda) was accepted for the ASCO 2026 annual meeting this May. Tusporsertib is a dual inhibitor that blocks both TGF-β and VEGF to improve the tumor microenvironment, offering new hope for patients unresponsive to existing immuno oncology drugs.
Given the remarkable 70.6% objective response rate (ORR) in previous trials, the additional data to be unveiled at the conference is expected to be a critical indicator for global licensing negotiations.
Another pillar, Merigolix, also produced overwhelming data in a European Phase 2a trial. The market was stirred by results showing that the high dose group’s pain score (NRS) was reduced to virtually ‘zero.’ TiumBio has already completed partial technology transfers to Daewon Pharmaceutical in Korea and Hansoh Pharma in China. Currently, the company is pursuing a “big deal” for the remaining rights in the North American and European markets.
Kim Hun taek CEO of TiumBio stated “The acceptance of the ASCO abstract is a result of global experts officially recognizing the value of Tusporsertib and Merigolix. We are currently in in-depth discussions with multiple global Big Pharma companies while accelerating development with the goal of commercialization by 2028.”
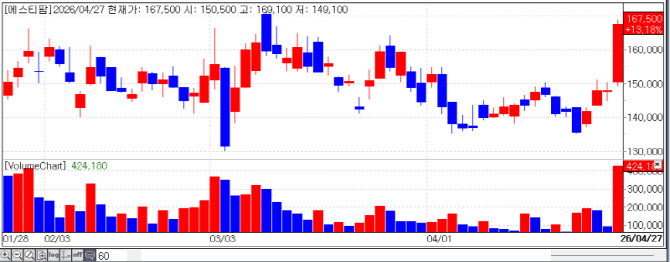 | | Recent stock price trend of ST Pharm. (Source: KG Zeroin MP DOCTOR) |
|
ST Pharm, ‘Oligo CDMO’ Operating Profit Explodes... “Factory Capacity Reaching Limit” ST Pharm ended the day at 167,500 won, up 13.18%. The catalyst for the surge was undoubtedly its “phenomenal performance.” ST Pharm reported a preliminary operating profit of 11.5 billion won for the first quarter, representing a staggering 1024.6% increase year on year. Revenue also rose 27.7% to 67 billion won, achieving both scale and profitability.
The primary driver of this turnaround is ‘Oligonucleotide,’ the core raw material for RNA therapies. Oligo raw materials are high margin items with significant manufacturing difficulty and high unit prices compared to general synthetic drugs.
Leveraging its world-class Oligo CDMO (Contract Development and Manufacturing Organization) competitiveness, ST Pharm has secured pipelines from global Big Pharma. With RNA therapies developed by companies like Ionis and GSK awaiting FDA approval this year and next, anticipation is reflected in the stock price that supply will transition from clinical scale to massive “commercial-scale” volumes.
ST Pharm‘s order backlog is also rising sharply. The Oligo division’s backlog, which stood at 204 billion won at the end of last year, surged about 70% in just three months to 340 billion won as of the end of March. Accordingly, the market expects ST Pharm to potentially expand production lines at its second Oligo plant, completed last September, ahead of schedule.
An ST Pharm official explained, “This strong performance is not merely due to currency effects but is the result of qualitative growth in our business structure, including an increased share of high-margin Oligo products. The RNA therapy market is just beginning to blossom, and as more pipelines enter the commercial stage, the demand for raw materials will increase exponentially.”
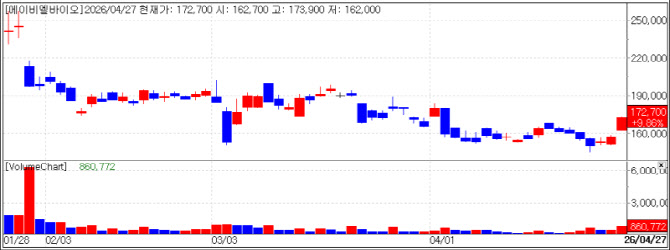 | | Recent stock price trend of ABL Bio. (Source: KG Zeroin MP DOCTOR) |
|
ABL Bio, Tech Transfer Success and ‘Next ADC’ Vision ABL Bio rose 9.86% to close at 172,700 won. As a leader in the domestic bispecific antibody field, the company received a significant boost from the Phase 2/3 clinical success of ‘Tobesevig,’ announced by its technology transfer partner, Compass Therapeutics.
Tobesevig, a candidate for biliary tract cancer, nearly doubled the median progression-free survival (PFS) compared to the control group and reduced the risk of disease progression by 56%. Since biliary tract cancer is a refractory cancer with extremely limited treatment options these results are evaluated to significantly increase the possibility of an FDA Biologics License Application (BLA). Separately, ABL Bio reaffirmed its technological edge by presenting its next-generation Antibody-Drug Conjugate (ADC) strategy at the recent AACR 2026 in San Diego.
The ADC market is currently shifting beyond simply attaching drugs to antibodies toward ‘Dual Payload’ competition, where two different drugs are attached. ABL Bio unveiled preclinical data for its bispecific ADC pipelines ABL206 and ABL209, demonstrating superior anti tumor effects and safety compared to conventional monospecific ADCs. The company plans to maximize its corporate value by directly conducting global clinical trials through its U.S. subsidiary, Neox Bio.
An ABL Bio official noted, “The ADC market paradigm is rapidly shifting toward payload (drug) competition. We will preempt the global market with our dual payload strategy that overcomes the limitations of existing topoisomerase based inhibitors.”
The surge in these three companies suggests a shift in the biotech investment landscape. The market no longer grants trust based on vague slogans like “developing a cancer cure,” simple name changes, or jumping on themes. Investors are now meticulously scrutinizing what data companies present at global conferences, how their actual revenue and operating profits are recorded, and how substantive their partnerships with global Big Pharma are.
Hong Soon-jae, CEO of BioBook, analyzed, “Companies like ST Pharm, which possess partnerships with large pharmaceutical companies or unrivaled manufacturing competitiveness, will gain even more momentum in an upward market. Conversely, companies that only put forward trendy keywords like ‘AI’ or ‘ADC’ without substantive research results will inevitably face selling pressure and diverge in performance.”




![Genome&Company Jumps on Clinical Momentum...ViGenCell Cools [K-Bio Pulse]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/04/PS26043000451b.jpg)



![지놈앤컴퍼니, 임상 본격화 기대감에 급등…숨고르기 들어간 바이젠셀[바이오 맥짚기]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/04/PS26043000447b.jpg)