Unauthorized reproduction or distribution is illegal and subject to criminal penalties.
Pharm Edaily enforces a zero-tolerance policy and will take strict action.
[Song Young-Doo, Edaily Reporter] Regulatory approval of a new drug and clinical data presented at a major global conference drove investor attention toward FutureChem and HLB Innovation. In contrast, ROKIT Healthcare extended its losing streak to 12 consecutive sessions amid growing concerns over core technology credibility and the use of R&D funds.
 | | FutureChem stock trend.(Source=KG Zeroin MP Doctor) |
|
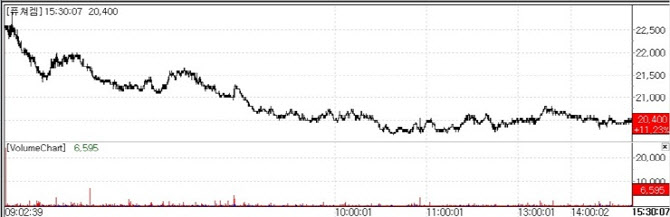
FutureChem Surges on 43rd Domestic New Drug Approval According to market data provider MP DOCTOR on May 4, FutureChem rose 11.23% to close at won 20,400. The company announced that its prostate cancer diagnostic radiopharmaceutical Prostaview Injection (Florastamin(18F)) has received marketing approval from the Ministry of Food and Drug Safety, becoming the 43rd new drug approved in Korea.
The product combines the positron emitting isotope fluorine-18(F-18) with a peptide targeting prostate-specific membrane antigen(PSMA). Following intravenous administration, PET-CT imaging enables precise visualization of tumor location and disease status.
Clinical efficacy was also confirmed. In a Phase 3 trial conducted across 11 domestic institutions, the primary endpoint positive predictive value(PPV) reached 86.96%, significantly exceeding the benchmark of 60.6%.
Compared to conventional imaging modalities such as CT, MRI, and bone scans(approximately 60.16%), the result represents an improvement of about 26.8 percentage points, highlighting its potential to reduce false positives.
With this approval, FutureChem has expanded its diagnostic radiopharmaceutical portfolio to four products, following FLT(lung cancer), PD View(Parkinson‘s disease), and AlzaView(Alzheimer’s disease).
A company official stated, “With this approval, we plan to accelerate both domestic supply and overseas expansion. Through collaboration with automated synthesis equipment firm Trasis, we have optimized production and established a foundation for global market entry, including China.”
The prostate cancer imaging market is considered a blockbuster segment, with over 400,000 PET-CT scans conducted annually in the United States alone. The global market is projected to reach approximately $13.16 billion by 2030, according to Grand View Research.
HLB Innovation Jumps on First Day of Trading Resumption HLB Innovation rose 13.85% to won 26,300 on its first trading day following a suspension due to a share merger process.
The rally was driven by its subsidiary Verismo‘s presentation at the AACR 2026, where interim Phase 1 results of its solid tumor CAR-T therapy SynKIR-110(STAR-101) were disclosed. The study was selected for a plenary session, the highest-profile presentation category at the conference.
SynKIR-110 is the first in human trial utilizing a KIR-CAR platform, targeting patients with advanced mesothelin-expressing solid tumors, including ovarian cancer, mesothelioma, and cholangiocarcinoma, who have relapsed or are refractory to standard therapies.
According to the abstract, no dose-limiting toxicities(DLTs) were observed through Cohort 3. Cytokine release syndrome(CRS) occurred in 33% of patients(3 out of 9), all of which were Grade 2 or lower, while no immune effector cell-associated neurotoxicity syndrome(ICANS) was reported.
In terms of efficacy tumor responses were observed in 4 out of 9 evaluable patients with tumor size reductions of up to 47%. One patient in Cohort 3 maintained a partial response(PR) for six months.
Janos Tanyi, Associate Professor at the Perelman School of Medicine and principal investigator of the trial, commented “SynKIR-110 has demonstrated a favorable safety profile and therapeutic potential without DLTs in the first three dose cohorts. The observed biological activity and disease stabilization in early stage trials are encouraging.”
Verismo also presented preclinical data on its hematologic CAR-T therapy SynKIR-310. The candidate, based on an NK cell derived multi chain KIR-CAR structure is designed to minimize T-cell exhaustion and sustain antitumor activity.
In animal models, SynKIR-310 showed superior antitumor efficacy compared to CD28- and 4-1BB-based CAR-T therapies, and was the only group to achieve 100% survival.
In an ongoing Phase 1 trial(CELESTIAL-301), a follicular lymphoma patient achieved complete response(CR) at the lowest dose level, with the response sustained for over six months.
 | | ROKIT Healthcare press release excerpt (Source=ROKIT Healthcare) |
|
ROKIT Healthcare Extends 12-Day Slide Amid Technology and Funding Concerns ROKIT Healthcare fell 6.54% to won 71,500, marking its 12th consecutive trading day of decline since April 16(won 103,200). The stock has nearly halved from its recent peak of around won 140,000 earlier this month.
The downturn is attributed to mounting concerns over the uncertainty of its regenerative platform technology and issues surrounding the use of funds raised from investors.
Earlier this month, the company released a press statement claiming a “complete hair regrowth in four weeks,” which briefly drove its stock up nearly 18% intraday. However follow up reporting revealed that the core patent remains at a provisional application stage, while key materials and mechanisms have not been disclosed. Preclinical data were also found to be limited.
The market has criticized the move as an “event-driven approach” that packaged unverified claims into definitive statements, with some raising concerns that it could escalate into a situation similar to Samchundang Pharm.
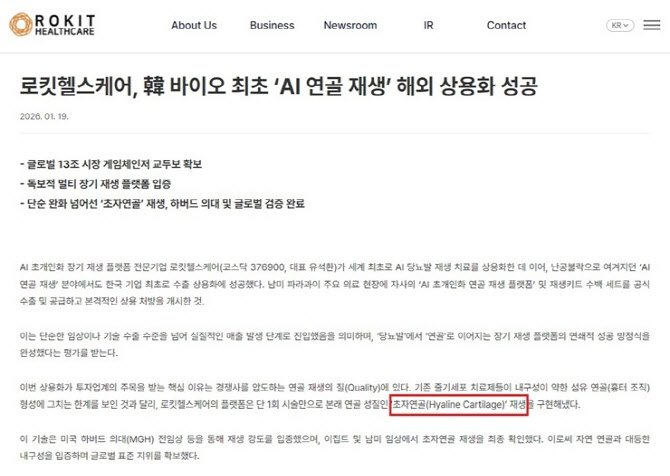
Questions have also been raised about the company’s cartilage regeneration platform. A review of cited studies indicated that the regenerated tissue was not true hyaline cartilage but rather “hyaline-like cartilage.”
In response, the company stated that “hyaline-like cartilage is a commonly used scientific term indicating functional cartilage regeneration.” However, inconsistencies were noted as the company had previously emphasized “hyaline cartilage” in its promotional materials.
Funding transparency has emerged as another key issue. Within less than a year of listing, ROKIT Healthcare raised over won 110 billion through its IPO(won 17.6 billion), convertible bonds (won 30 billion), and a rights offering(won 62.5 billion), all earmarked for regenerative R&D.
However, its 2025 business report showed that only about won 800 million(approximately 10%) of the convertible bond proceeds had been spent, while the entire IPO proceeds were allocated to “operating expenses and others.”
Industry observers noted that “the combination of unverified core technology and opaque fund utilization has significantly undermined investor confidence.”




![로킷헬스케어 1.9조 수주 계약이라더니…구속력 無·매출 전환율 0.8%[only 이데일리]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/05/PS26050400118b.jpg)